  
They are also called synthetic elements and have radioactive properties. Some elements are not found in nature but are synthesized in laboratory settings. They are used for coinage, jewelry, and alloys and have different commercial applications. Other precious metals include osmium, ruthenium, silver, and rhodium. The group of precious metals includes elements such as palladium, ruthenium, platinum, iridium, and gold. CERN researchers suggest that its isotopes can be used in cancer treatment therapies. The rarest element is astatine, and scientists estimate that the total amount found is less than 1 gram. Ununoctium is heavier and the heaviest known chemical element, but it is manmade. Uranium is the heaviest element that occurs freely in nature. Hydrogen has important commercial applications, for example, hydrogen fuel cells and the manufacture of chemical products. Hydrogen is the lightest element, and it is also the most abundant one. Francium, however, is the most expensive element that can be produced. Lutetium is a metal and the most expensive chemical element available. Periodic Table Curious Facts The Most Expensive Element 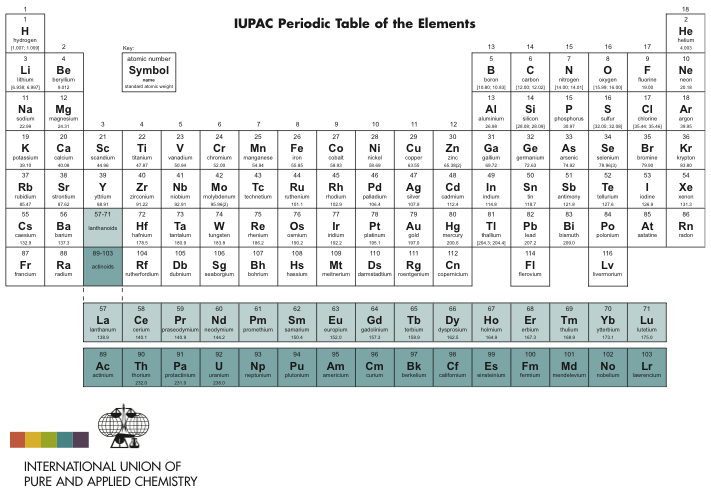
Metalloids share properties with both nonmetals and metals. Metals are malleable, shiny, and ductile while nonmetals lack metallic properties and are volatile. The major categories are metalloids, nonmetals, and metals, and most elements in the Periodic Table are metals. The s block contains alkaline earths and alkali metals. The p block includes post-transition metals, semimetals, and nonmetals, with the exception of helium and hydrogen. The f block includes inner transition elements and the d block is made of transition elements. There are 4 blocks in the Periodic Table - f, d, p, and s. Blocksīlocks combine adjacent groups and are also called element families. Many period 6 elements are toxic, heavy, and radioactive. The rare earth elements are found in period 6. Period 1 contains two elements, helium and hydrogen while period 7 contains radioactive elements. There are 7 periods of elements that group elements with similar properties. The Periodic Table groups elements into the cobalt, chromium, vanadium, scandium, copper, cobalt, and other groups. The noble gases and halogens are in groups 18 and 17, respectively. 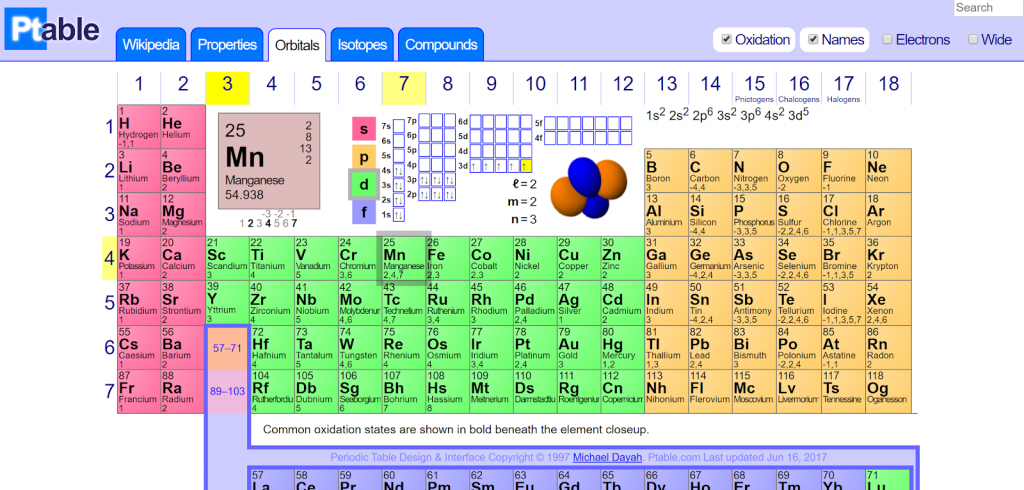
Group 1 of the Periodic Table groups together the alkali metals while group 2 contains all alkaline earth metals. The so called typical elements are found in the first two rows. The Periodic Table includes 18 groups, and each group contains elements with similar chemical and physical properties of the outermost electron shells. Attempts were made to synthesize several new elements, including unbiseptium, unbihexium, unbiquadium, and unbibium. According to the American scientist Glenn Seaborg, element 130 is the highest possible. It is still to be discovered how far the Periodic Table of Elements extends. The elements were organized based on their physical and chemical properties. It was only in 1869 when Dmitri Mendeleev, an inventor and chemist of Russian origin, discovered the Periodic Law and organized all chemical elements in columns and rows. Johan Dobereiner grouped chemical elements into triads, and De Chancourtois formulated a chart with closely related elements. However, the first attempts at systematization occurred in 18. Early Systematization AttemptsĪ total of 63 elements have been discovered by 1869. It is known as the first scientific discovery of a chemical element. Phosphorus became the first element to be discovered by Hennig Brand in 1649. The Periodic Table displays all known chemical elements which are grouped by chemical properties and atomic structure.Ĭopper, silver, gold, mercury, tin, lead, and other elements have been known since ancient times and were used to make jewelry, coins, and tools. Origins of the Periodic Table of Elements Dynamic Periodic Table H 1 Hydrogen 1.0079 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
